Medical Equipment Supplier China | Reliability & Quality Systems: From Early Iteration to Export-Ready Reliability (A 15-Year View)
Medical Equipment Supplier China | Reliability & Quality Systems: From Early Iteration to Export-Ready Reliability (A 15-Year View)
China’s Western-style medical device manufacturing started relatively late compared with long-established markets. But the pace of progress over the past decades has been substantial.
From my perspective, China’s industry has evolved like a bare concrete apartment gradually becoming a well-built, fully furnished home: the structure becomes stronger, the details become more refined, and the overall experience becomes more dependable year after year.
After 15 years working in medical devices, I have seen three major phases: early engineering-driven iteration, supply-chain and tooling expansion, and the current stage where more manufacturers are building export-ready systems around quality, documentation, and service.
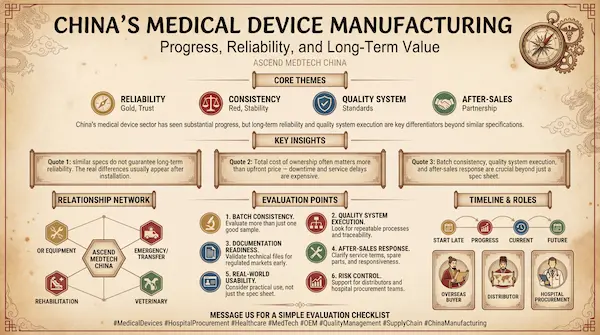
This article shares practical observations for overseas distributors and hospital procurement teams who want to understand what has changed and how to evaluate suppliers today.
1) The early stage: building without a mature ecosystem medical equipment supplier China
In the early stage of China’s industry development, many factories did not have mature molds, standardized components, or a stable supporting supply chain. Product development relied heavily on hands-on engineering and incremental improvement.
Take operating lights as a representative example. In the beginning, many products were built with limited tooling and limited component standardization. Designs often followed what the market had already validated, and manufacturing maturity improved step by step through repeated iteration.
This phase was not “perfect,” but it was necessary: every manufacturing ecosystem needs a period of learning by building.
2) From “available” to “reliable”: specs are not the full story of medical equipment supplier China
As more companies entered the market, tooling improved, molds became more common, and the component ecosystem began to form. LED technology became more accessible, and product performance improved in many categories.
However, the market also learned an important lesson: a product can look similar on paper, but long-term reliability depends on engineering depth and systems discipline.
For overseas buyers, the real differences often show up in:
- batch consistency (not just one good sample)
- thermal management and stability over time
- quality control execution (not just “QC wording”)
- documentation and traceability
- after-sales response, spare parts, and clear service terms
3) Volume created cash flow, and cash flow financed upgrades of medical equipment supplier China
Over time, procurement volume increased. With more orders and more stable cash flow, manufacturers could invest in:
- stronger processes and tooling
- better supply chain partners
- improved testing capability
- clearer technical documentation
- more structured service and feedback loops
This is also why a number of Chinese companies started to build international recognition through long-term investment and market validation. In parallel, the overall industry became more segmented: some suppliers remained price-driven, while others moved toward higher consistency and export readiness.
4) Export markets value different things — and China’s role is expanding of medical equipment supplier China
In many regions, medical devices must balance clinical needs with limited budgets. That practical reality has created space for Chinese manufacturers to support healthcare access in a way that is economically feasible.
In our experience, different markets often emphasize different priorities:
- Southeast Asia: overall value and practicality
- some developing regions: affordability and availability
- Eastern Europe: increasing focus on consistency, craftsmanship, and durability
- more regulated markets: documentation readiness, compliance, and stable long-term performance
China is not “one quality level.” The more important trend is that a growing number of suppliers are moving from exporting products to exporting capability: quality systems, documentation, and credibility Medical Equipment Supplier China
Practical guidance for overseas buyers
1) If you are a distributor: factory or trading company?
There is no single correct choice. It depends on your business model.
If local registration is required, working directly with a qualified manufacturer is often the most straightforward route. Key checks include:
- technical file completeness and consistency
- certificates and regulatory readiness for your market
- traceability of critical materials and components
- agency scope and exclusivity terms clarified before contract signing
If you handle complex hospital projects across multiple categories, a specialized supplier can reduce risk and workload through:
- cross-category product selection and project coordination
- supplier screening and quality checks
- after-sales management and faster response
- experience matching specifications with real-world use scenarios
This is the type of work we do at ASCEND MEDTECH CHINA: helping overseas partners choose the right configuration and manage the hidden risks that often appear after installation and months of use.
also need to follow standard :ISO 13485:2016/ IMDRF:Quality Management Systems
2) If you are a hospital procurement team: focus on total cost of ownership
Hospitals often need to balance Medical Equipment Supplier China:
- upfront purchase price
- reliability and downtime risk
- service response and spare parts availability
- training, maintenance, and replacement cycles
Some suppliers can offer very competitive pricing, but may have limited export experience or weaker service structures. A supplier with category expertise and transparent risk communication can reduce hidden costs related to maintenance delays, repeated repairs, or early replacement.
3) What we learned from factory-side comparisons: differences are often “invisible”
In many product categories, price differences usually come from what you cannot see immediately:
- material grade and structural strength
- thickness and reinforcement design
- process discipline and test flow
- documentation repeatability and traceability
- service terms and spare parts planning
Examples that often require deeper evaluation:
- operating tables: real load stability under testing conditions, not only a nominal number
- operating lights: CRI stability, thermal management, and long-term consistency
- ceiling pendants: joint stability and gas terminal standards
- wheelchairs: battery compliance and full test verification
- stair chairs: safety design and true one-person operability in real scenarios
In short: without deeper evaluation, risks are easy to miss early.
4)A practical (non-exhaustive) view of medical equipment supplier China clusters
One question overseas buyers often ask is: “Where are medical devices mainly made in China?”
There is no perfect map, and the distribution is not absolute. However, from what we have seen in the field, medical equipment supplier China ecosystem often forms regional clusters shaped by history, supply chain availability, talent pools, and local industrial policies.
A simplified overview (for reference only):
- Shanghai: more established OR-related companies and higher-tech development, including parts of the IVD ecosystem.
- Jiangsu: stronger presence in emergency-related products and respiratory equipment in certain segments.
- Shandong: notable concentration in OR equipment such as operating lights, operating tables, and hospital trolleys/carts.
- Hebei: more price-competitive segments such as basic hospital bed products (with quality varying widely by supplier).
- Guangdong: active in rehabilitation equipment across multiple categories.
- Shenzhen: stronger in electronics-driven medical devices and related components.
- Zhejiang: broad activity in medical consumables and supporting supply chains.
For buyers, the key takeaway is not “which province is best,” but how to match the right supplier cluster with your requirements—especially documentation readiness, batch consistency, and after-sales execution.
Final thoughts
Medical Equipment Supplier China device manufacturing is improving, but it remains a segmented market. The direction, however, is clear: stricter regulation, tougher competition, and more suppliers building export-ready systems.
Our belief is simple: overseas buyers should be able to purchase with confidence — through better product understanding, transparent risk control, and reliable support. This is the long-term value we aim to deliver at ASCEND MEDTECH CHINA.
However, we will continue to strive, continuously enhancing the product’s craftsmanship and quality reliability, aiming to compete with European and American equipment, ensuring our dealers can be confident and the hospitals that purchase our products can also feel assured.



